3 | Molecular chemistry#
Sources#
Slides#
Deck 1 | Hydrocarbons#

Slides (pdf)
Deck 2 | Functional Groups#

Slides (pdf)
Deck 3 | Coordination Chemistry#

Slides (pdf)
Deck 4 | HSAB Theory#
Slides (pdf)
Deck 5 | Crystal Field Theory#

Slides (pdf)
Deck 6 | Ligand Field Theory#
Slides (pdf)
Deck 7 | Tetrahedral Ligand Fields#
Slides (pdf)
Deck 8 | Absorption Spectroscopy#

Slides (pdf)
Learning Objectives#
Organic Chemistry#
Drawing and naming hydrocarbons
Draw and understand the equivalence of lewis structures, line angle structures with hydrogens, and line angle structures with implicit hydrogens
Know the naming conventions of the simple linear alkanes (ethane, propane, butane, …) and the group names (ethyl, propyl, butyl, …)
Know the basic naming conventions of alkanes with branches
Find and number the longest chain in a simple hydrocarbon identify the group names for the branches and their carbon index along the longest carbon chain
Know the basic naming conventions of alkenes and alkynes
Know the structure of benzene and simple cyclic alkanes (cyclopropane, cyclopentane, cyclohexane, …)
Be able to number cycloalkanes and not the position of branched groups (e.g. 1,2-dimethyl-cyclohexane)
For alkenes be able to draw both the cis and trans isomers when possible and understand when there is not difference between cis and trans.
Be able to label the degree of a carbon (primary (1º), secondary (2º), tertiary (3º), quaternary (4º))
Isomers#
Isomers are 2 or more molecules with the same chemical composition (stoichiometry) where the atoms are arranged differently with respect to each other.
Be able to identify if compounds are:
structural isomers (same composition, difference connectivity)
geometric isomers (same connectivity, difference arrangement in space (e.g. cis vs trans))
optical isomers (enantiomers)
completely different compounds (different chemical compositions)
Organic Reactions#
Be able to identify a substitution reaction
Be able to determine the products of a substitution reaction given the reactants and the type of reaction
Be able to identify an elimination reaction
Be able to determine the products of a elimination reaction given the reactants and the type of reaction
Be able to identify an electrophilic addition reaction
Be able to determine the products of a electrophilic addition reaction given the reactants and the type of reaction Dehydrohalogenation = a specific type of elimination reaction hydrohalogenation = a specific type of electrophilic addition reaction reaction
Be able to identify an electrophilic substitution reaction at a benzene ring
Be able to determine the products of a electrophilic substitution reaction given the reactants and the type of reaction
Be able to identify a condensation reaction (e.g. formation of an ester/amide bond)
Be able to determine the products of a condensation reaction reaction given the reactants and the type of reaction Resonance
Resonance#
Be able to draw the resonance structures give a molecules structures
Determine if the resonance structure you drew are equienergetic (chemically equilvalent) or not equienergetic.
Understand how resonance can make certain functional groups “electron withdrawing” (i.e nitrobenenze) or “electron donating” (i.e. phenol) and how that may influence the products or rate of a reaction.
Conjugation and aromaticity#
Identify and count the number of \(\pi\) bond that are “conjugated” in a molecule
Understand the approximate relationship between the extent of conjugation and the color of a hydrocarbon.
Determine if a molecule is aromatic (cyclic, \(4n+2\) \(\pi\) electrons), anti aromatic (cyclic, \(4n\) \(\pi\) electrons), or neither (either non cyclic or the \(\pi\) electron count is not equal to \(4n\) or \(4n+2\)) where \(n = 0, 1, 2, 3, 4, ...\)
Functional groups#
Be able to identify and name compounds with the following functional groups fluoro, chloro, bromo, iodo, hydroxy, ether, phenol, aldehyde, ketone, carboxylic acid, ester, amine, amide, alkene, alkyne, phenyl
Draw dipoles on functional groups and locate atoms with partial positive or partial negative charges
Identify nucleophilic and electrophilic regions of molecules
Understand the basic reactions of the different functional groups
haloalkanes: nucleophilic substitution
alcohols: acid base chemistry, oxidation to aldehyde/ketones, nucleophilic substitution with haloalkanes to form ethers, condensation with carboxylic acid to form esters
Synthesis of ethers from alcohols and haloalkanes
Synthesis of aldehydes via oxidation of primary alcohols
Synthesis of ketone via oxidation of secondary alcohols
Synthesis of esters from carboxylic acid and alcohol
Synthesis of peptides from condensation of amino acids
Some basic properties of each functional group
Haloalkanes are polar covalent bonds and good “building block” molecules
Alcohols are usually very weak acids (weaker than water) and participate in hydrogen bonding. Their conjugate bases (alkoxides) are excellent nucleophiles (make ethers and esters)
Ethers are volatile and the key functional group of the industrially important polyethylene glycol
Phenols are more acidic that other alcohols due to resonance structures and the delocalization of electrons within the benzene ring. Used to make important resins, flavor molecules and pigments.
Aldehydes and ketones are oxidized alcohols, the have a strong dipole (polar) along the C=O bond, and the carbon is trigonal planar. They are often found in flavors and fragrance compounds
Carboxylic acids are weak acids (pKa’s often around ~4-5). Acids taste sour and are often pungent (e.g. oxidized fats = rotten). Many are described as onerous, foul, sometimes sharp (like vinegar)
Esters are also often found within flavors and fragrances. They are formed by condensation reactions of carboxylic acids and alcohols (these compounds are often sweet or fruity)
Amines are weak bases (pKa ~10). The also smell sharp or often fishy.
Amino acids are not really functional groups so much as building block molecules of proteins that contain both an amine and a carboxylic acid group
Amides are like esters but formed with an amine instead of an alcohol. While the flavor chemistry of umami is more complicated than others typical flavor molecules often include amide bonds.
Coordination Chemistry#
Ligands and the Dative Bond#
Communicate the difference between a dative bonding interaction and other types of bonding like covalent, ionic, and hydrogen bonding.
Fundamentals or lewis acid and lewis base chemistry and thinking or very polar covalent bonds as a pair of electron donor and electron acceptors
Know the names of common ligands, their charge, and the number of electron pairs that donate to a metal center
Given the structure of a ligand predict how electron pairs it can donate to a metal center and draw the chemical structure of the ligand coordinated to the metal
Explain why chelate structures are more stable the similar structures without a chelate ligand
Determine the number of d electrons a metal center has based on its formal oxidation state
Determine the total number of electrons for a metal center based on the number of d-electrons and the number of electrons donated to the metal center by ligands
Identify geometric, structural, and optical isomers within tetrahedral and octahedral metal complexes.
Be able to draw different isomers of a metal complex given at starting compounds structure
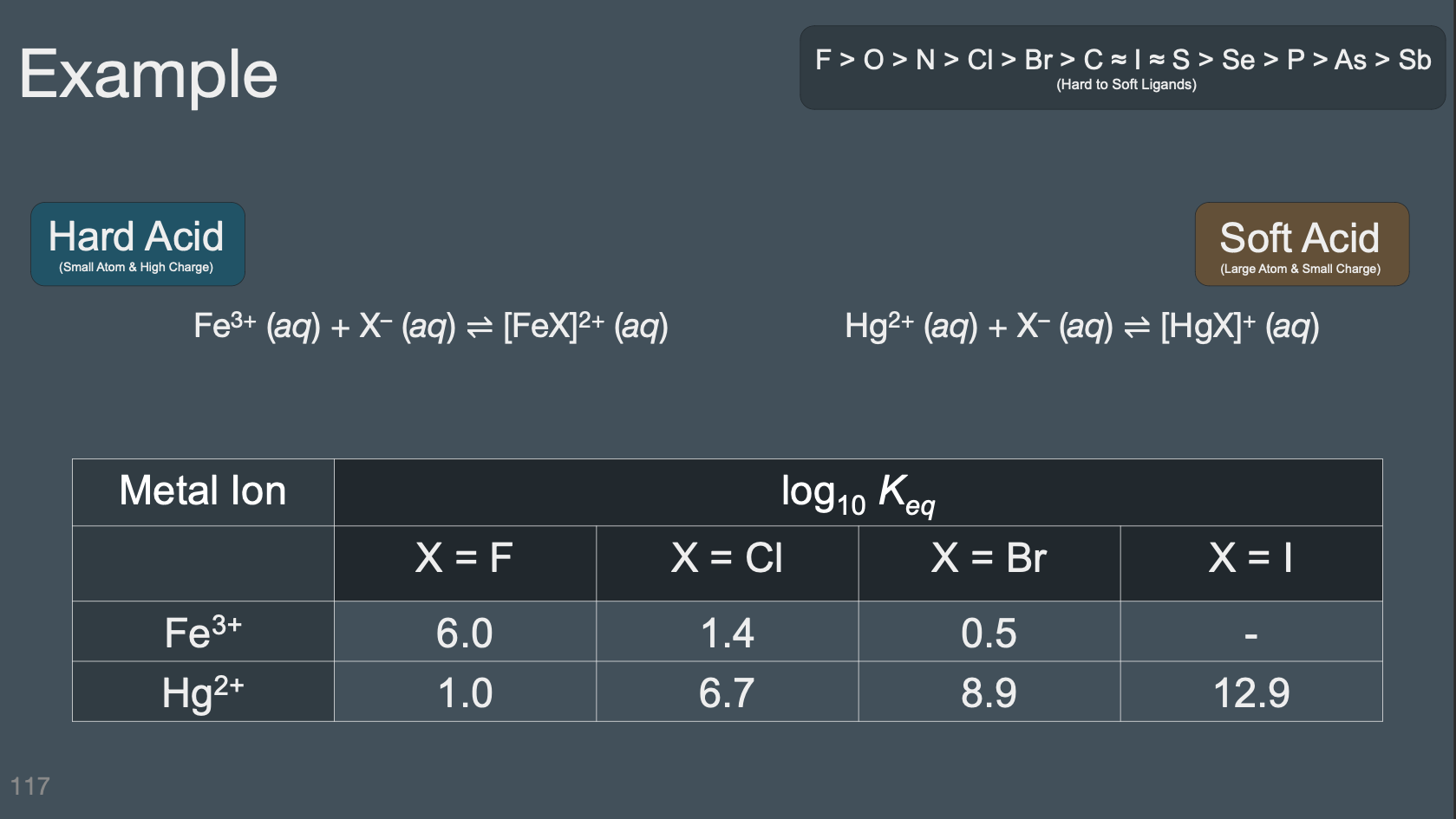
HSAB Theory#
Determine if a given ligand is going to be a hard or soft Lewis Base
Determine if a given metal ion is going to act as a hard or soft Lewis Acid
Given two pairs of coordinated Lewis acid-base pairs, predicted which one will have the strongest interaction based on possible Hard-Hard, Soft-Soft, and Hard-Soft interactions
Sort acids and bases on a spectrum from hard to soft
Crystal Field Theory#
Draw and label the 5 d-orbitals in their correct orientations
Communicate how the d-orbitals’ energies change going from a free ion to a spherical crystal field and then after distorting the spherical field to a lower octahedral symmetry
Draw the orbital diagram for the d-obital splitting in an octahedral field
determine the number of d electrons in a complex give the molecular formula
Correctly fill the d-orbitals with electrons following the Aufbau Principle and Hunds rules for BOTH the High Spin (HS) scenario and the Low Spin (LS) scenario
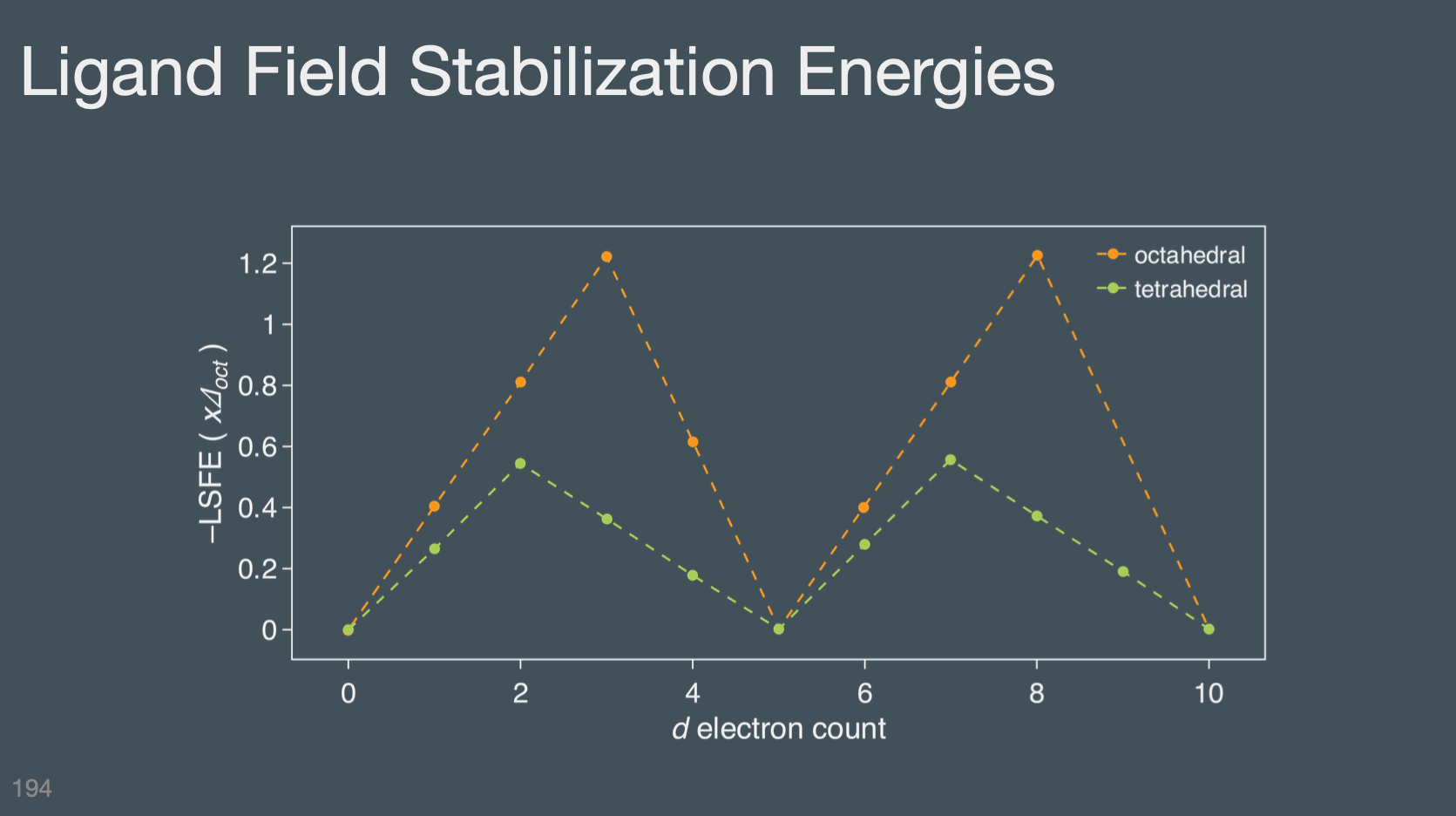
Calculate the CFSE for an octahedra complex
Determine the total spin (S) for the molecular by adding the component ms values of the d-electrons
Predict based on your orbital splitting diagram if a compound will be diamagnetic or paramagnetic
Use the spectrochemical series to qualitatively predict \(\Delta_{oct}\) and predict if you may expect a metal center to be high spin (HS) or low spin (LS)
Know the d-orbital splitting pattern (2,3) for tetrahedral and that \(\Delta_{tet}\) is always smaller than \(\Delta_{oct}\) (\(\Delta_{tet}= \frac{4}{9} \Delta_{oct}\))
Tetrahedral metal complexes will always be High Spin for the 1st row transition metals
2nd and 3rd row transition metals are always Low Spin regards of the shape of the ligand field.
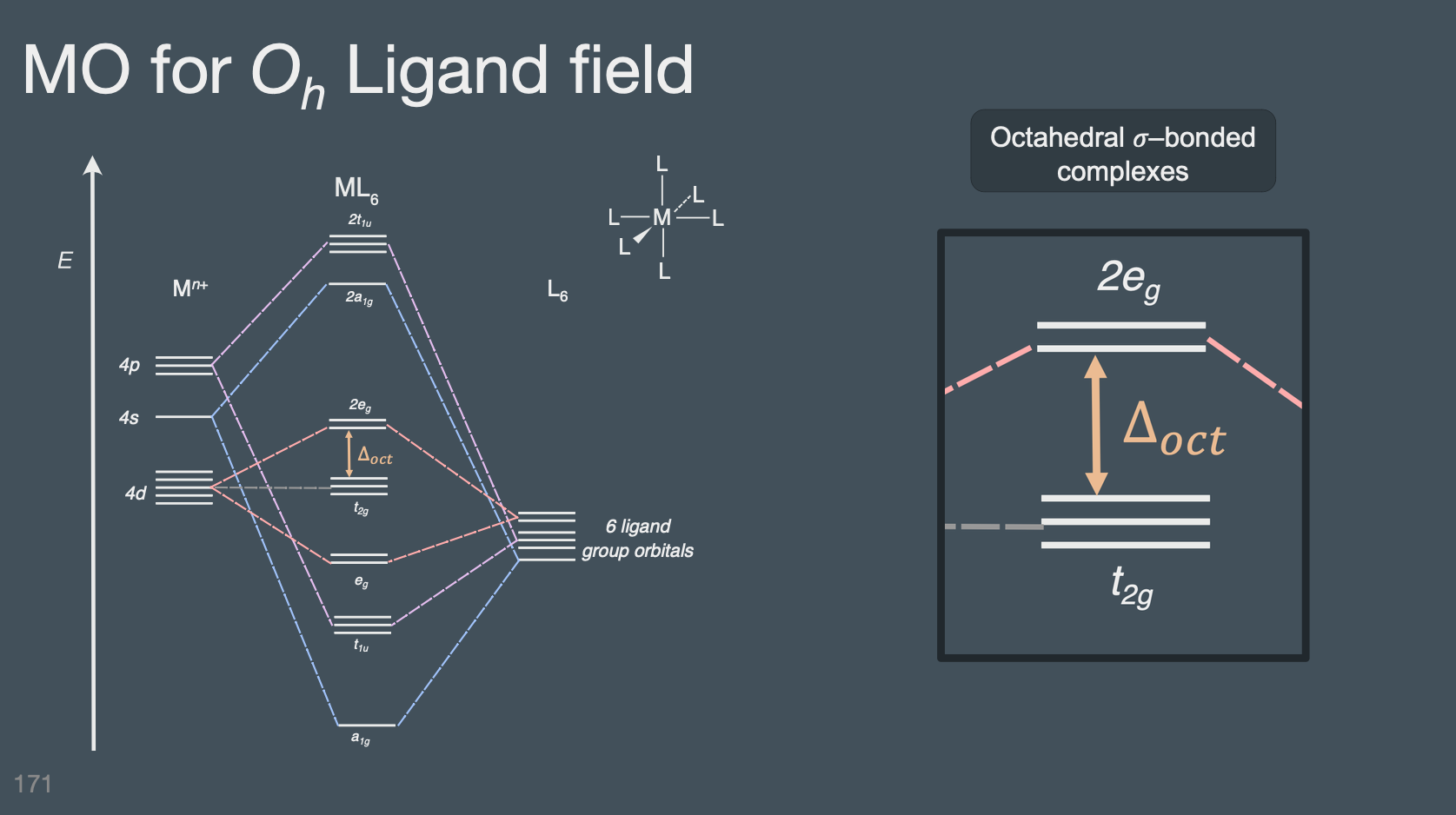
Ligand Field Theory#
Communicate the origins of ligand \(\pi\) donation (pi base) and why it decreases \(\Delta_{oct}\) Predict if a ligand structure will participate in ligand \(\pi\) donation (i.e. look for additional lone pairs (more the one) on the donating ligand atom)
Communicate the origins of ligand \(\pi\) acceptor (pi acid) interactions and why it increases \(\Delta_{oct}\)
Predict if a metal center, based on it’s d-electron configuration, can participate in \(\pi\) backbonding (bonding interaction with ligand \(\pi\)-acceptor)
Predict if a ligand (given its structure) will participate in ligand \(\pi\) acceptor interactions with a metal center (i.e. look for double bond in the ligand near the metal center)
Draw a sigma bonding interaction (cartoons of the orbital shapes) between a ligand p-orbital and either a \(d_{z^2}\) or \(d_{x^2-y^2}\)
Draw a \(\pi\) bonding interaction (cartoons of the orbital shapes) between a ligand p-orbital and one of the dpi orbitals: \(d_{xz}\) , \(d_{yz}\) , \(d_{xy}\)
Do so for a ligand \(\pi\) donor
Do so for a ligand \(\pi\) acceptor (metal \(\pi\) backbonding)
Absorption Spectroscopy#
Use the different units and measure of monochromatic light (wavelength, photon energy, frequency, wavenumber) (nm, eV, cm–1)
Communicate how we observe color and how molecules interact with light reflected off a surface to display coloration
Communicate how we observe color and how molecules interact with light transmitted through a clear solid or solution (Beer’s Law)
Colors get darker as the concentration of absorbing molecules in solution increases and the path light travels through solution increases
For metal complexes explain and predict how a spectrum will change with an increase and decrease in d-orbital splitting
As above you should be able to predict if a \(\Delta_{oct}\) will increase or decrease if you change the metal ion’s charge, the metal ions identical, or the ligand identity (i.e. spectrochemical series)
Given a UV-vis absorption spectrum predict the color of a compound
Communicate how all compounds tend to absorb light in the ultraviolet range of the electromagnetic spectrum